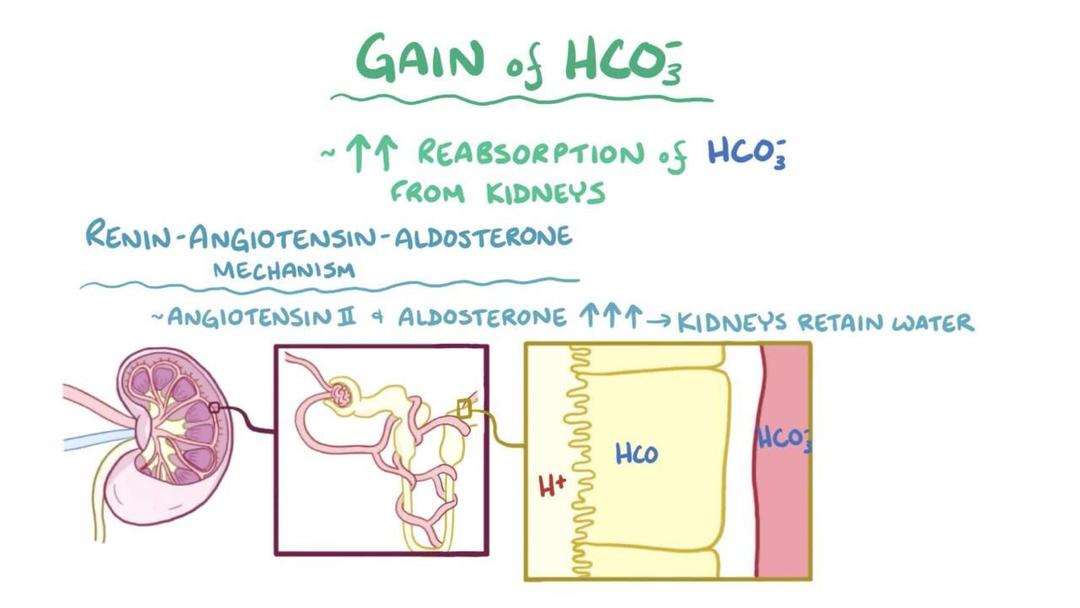
Overview of Metabolic Alkalosis
Metabolic Alkalosis (https://www.youtube.com/watch?v=eTOFH8B_Rsg&list=PLY33uf2n4e6PT53f0Z5LmFHo7Vb0ljn5b&index=11) by Osmosis (https://open.osmosis.org/) is licensed under CC-BY-SA 4.0 (https://creativecommons.org/licenses/by-sa/4.0/).
In these topics