Courtesy of Dr. Dawn Boothe.
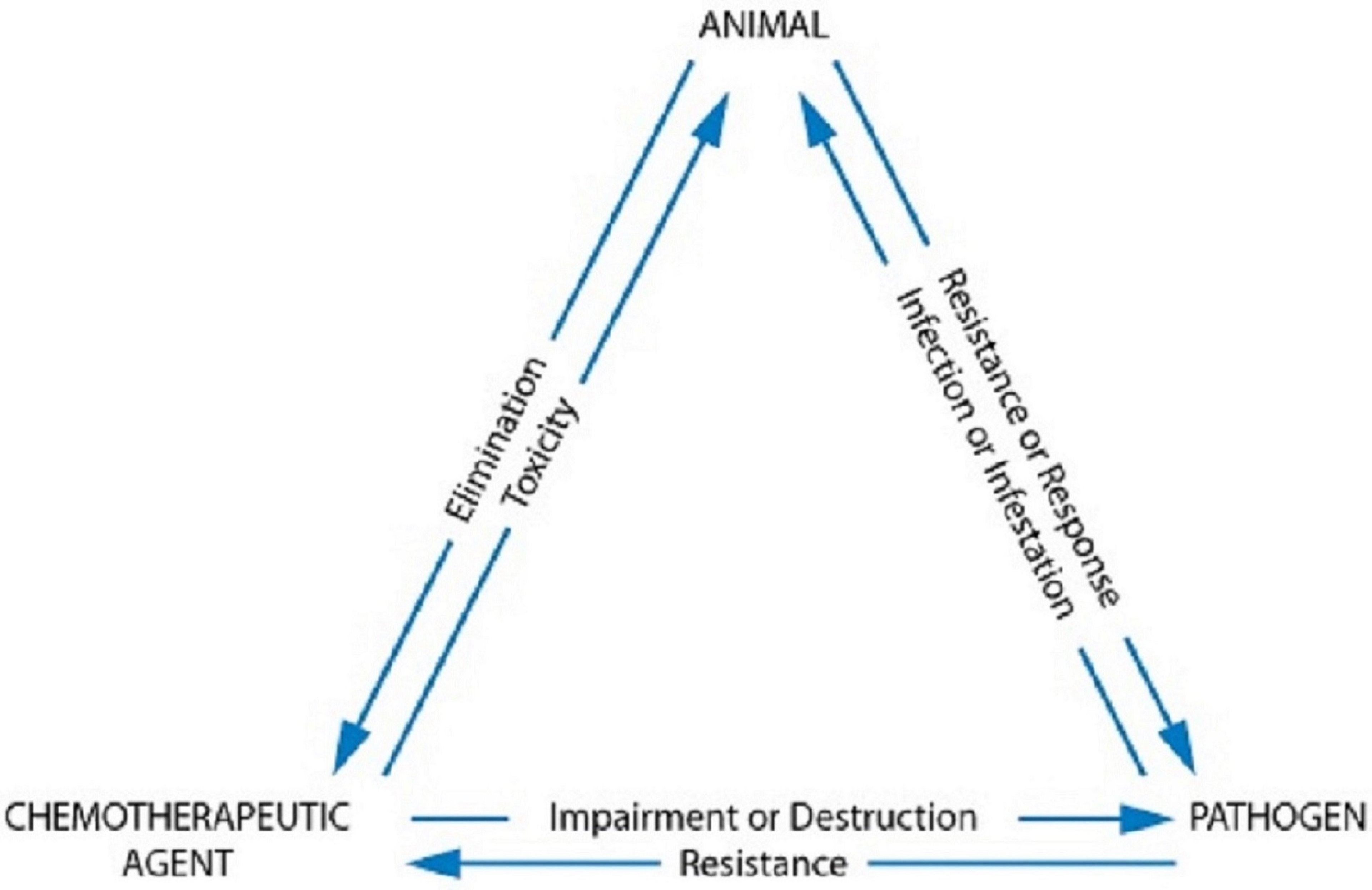
Treatment with chemotherapeutic agents involves complex interrelationships among the host animal, infecting pathogen, and drug, the interactions of which comprise the chemotherapeutic triangle. Whenever a drug is used with the intent to kill the target, multiple factors affect therapeutic success. For instance, the target organism will implement mechanisms to avoid harm—most notably, resistance mechanisms. Microbes in particular are associated with numerous virulence factors and other mechanisms for self-protection, often also making the host sick at the same time. Likewise, host factors, which seemingly should help the host fend off infection, can become detrimental to successful treatment.
For most infections, successful treatment depends on the host having a functional immune system. Drug factors also influence the success of treatment, most notably because drugs must be able to penetrate the site of infection to successfully kill the microbe. These are the interactions between the three points of the chemotherapeutic triangle: host, pathogen, and drug. If the goal of antimicrobial treatment is simply to resolve clinical signs of infection, then attention may not be given to the critically important aspects of treatment, such as toxicity or mitigating future resistance.
In general, use of an antimicrobial decreases the size of the susceptible population of the infecting pathogen; however, it is likely to leave behind a residual, yet resistant, population. Immunocompetent animals are likely to eradicate this residual population, but at-risk animals may not. Therefore, the population may regrow to a size capable of once again causing clinical signs (eg, 103–105 CFUs in the urinary tract).
Much discussion in this chapter is intended to support the design of dosing regimens (appropriate choice of drug, dose, and interval) that maximize antimicrobial efficacy while minimizing the risk of emergent antimicrobial resistance. This discussion of chemotherapeutic agents focuses on drugs that combat infecting bacterial pathogens; drugs used against fungal and viral pathogens are discussed in Antifungal Agents and Antiviral Agents, respectively.